Background
The Board of Gavi, the Vaccine Alliance is meeting in Delhi this week to review and decide future investments, including funding for the establishment of a global emergency stockpile of an Ebola vaccine that has just been quality assured by the World Health Organization.
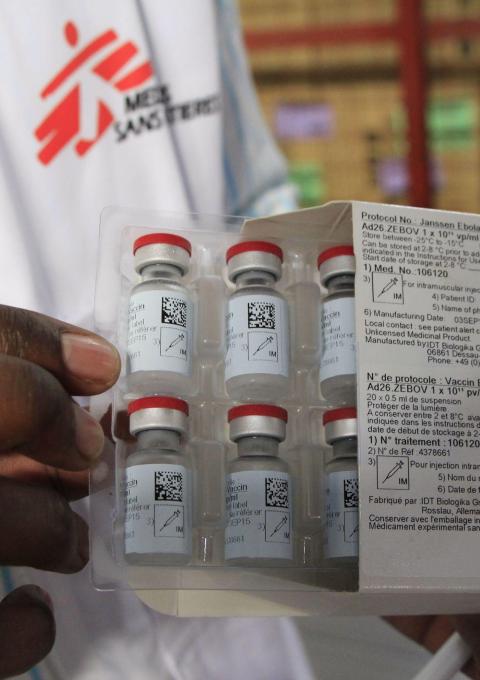
Merck was granted conditional approval by the European Medicines Agency (EMA) for its Ebola vaccine, rVSV-ZEBOV on November 11th and the vaccine was subsequently ‘prequalified’ by WHO, paving the way for procurement of this vaccine by UN agencies.
So far, more than 250,000 people have been vaccinated with rVSV-ZEBOV, which is reported to be 97.5% effective at preventing the disease. Despite of this vaccination campaign, the outbreak remains a serious public health concern. In September, MSF called for an international, independent committee to evaluate and extend the strategy and manage vaccine supplies more transparently.
Jason Nickerson, Humanitarian Affairs Advisor, MSF:
Gavi’s announcement that it will finance the establishment of an emergency stockpile of 500,000 doses of Ebola vaccine is a positive step. The use of the International Coordinating Group (ICG) mechanism has been agreed in order to ensure transparency and good governance for the allocation and management of this Ebola vaccine stockpile. The ICG mechanism, that works through the World Health Organization (WHO), already exists to manage emergency stockpiles of meningitis, cholera and yellow fever vaccines.
The rVSV-ZEBOV vaccine is the result of a collaborative research and development effort by public and philanthropic entities, not by Merck alone. MSF calls for a thorough, independent, and transparent estimate of the costs of manufacturing the vaccine, to ensure that the price paid is fair and reasonable.
MSF also calls for complete transparency in the overall global stockpile available to respond to Ebola outbreaks. As additional emergency stockpiles are in the process of being established, including in the United States, there is also an urgent need to have a mechanism guaranteeing that all national stockpiles can be swiftly pooled in case they are needed to broaden response capacity at a global level in any future outbreaks. This critical vaccine needs to be available when and where it’s needed in future Ebola outbreaks, to allow public health teams to respond quickly and effectively to protect the lives of people who are affected.”