Deborah Waterhouse
CEO, ViiV Healthcare
United Kingdom
cc Bill Collier, Head of Pediatric Strategy
cc Helen McDowell, Director of Government Affairs, Access & Patient Advocacy
Dear Mrs Waterhouse,
We are writing to express our concerns about the on-going delays in making the key pediatric HIV medicine dolutegravir available. On November 17, 2017, the global HIV community welcomed the commitment from ViiV at the Rome Pediatric HIV meeting, to make dolutegravir pediatric formulations available at the cost of production in low and middle-income countries until generics become available, but we have yet to see concrete steps to ensure this will happen.
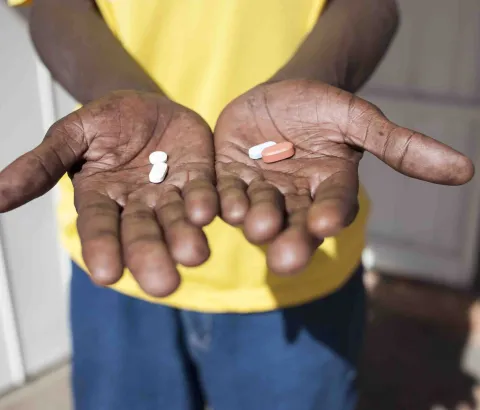
The vast majority of the 2.1 million children living with HIV do not have access to life-saving treatment or continue to receive suboptimal treatment. These children would benefit from dolutegravir-based regimens that have fewer side effects and a stronger barrier to resistance.
However, despite ViiV’s public promise, there has been little advancement to date, a full eight months later, to make this promise a reality. ViiV has barely started filing for registration of its available pediatric formulations, with dossiers submitted in only three countries in sub-Saharan Africa. This is particularly problematic because ViiV refuses to sell pediatric dolutegravir to countries where the medicine is not yet registered, even though the use of import waivers is a common practice accepted by most pharmaceutical companies for importation of unregistered ARVs into countries where they are needed.
Given the preference from the Pediatric ARV Drug Optimization group for dolutegravir as the best option for children and their recommendation to move towards alignment with adult treatment options, it is expected that the next World Health Organization HIV guidelines update will include dolutegravir as part of first-line treatment for children living with HIV. However, dolutegravir dosing is still not available for younger children; pediatric formulations are not available in countries where they are needed, and pediatric 5 mg dispersible tablets and the fixed-dose combination of ABC/3TC/DTG do not exist at all. As a result, 40% of children who are on HIV treatment continue to receive sub-optimal nevirapine-based regimens, putting them at risk of increased side effects, resistance and treatment failure, thereby increasing their risk of contracting opportunistic infections such as tuberculosis (TB), and dying.
ViiV must commit to take immediate, concerted actions in order to uphold its 2017 promise. We, the undersigned, demand that ViiV undertake the following actions, in order to ensure access to affordable pediatric formulations:
- R&D - Expedite finalization of dosing studies for younger children to allow simplified dosing for Lower- and Middle-Income Countries and the development/registration of 5mg dispersible tablets.
- REGISTRATION - start filing dossiers for existing pediatric dolutegravir for 10 mg and 25mg tablets, prioritizing countries with high HIV-prevalence, using WHO’s Collaborative registration process for participating countries. ViiV should subsequently publish the registration status (both filed and approved) by country online.
- PROCUREMENT – Make dolutegravir 10 mg and 25 mg tablets available for procurement by countries where registration is not complete by using importation waivers for unregistered medicines.
We call on ViiV to urgently take these steps and revise its policies and practices in order to allow countries to purchase and use pediatric dolutegravir formulations as quickly as possible.
If ViiV follows these actions, it will signal its stated dedication to helping improve the lives of ALL people living with HIV.
We look forward to your response by the eve of the International AIDS Conference, along with timelines for the actions outlined above.
Sincerely,
Access to Rights and Knowledge (ARK) Foundation
Action Against AIDS Germany
African Services Committee
Afro Global Alliance Ghana
AIDS Access Foundation
AIDS Healthcare Foundation
AIDS and Rights Alliance for Southern Africa (ARASA)
Association HIV.LV, Latvia
Coordinadora estatal de VIH y sida (CESIDA)
Club Kviten, Ukraine
Coalition PLUS
Consortium of Key Communities of Ukraine
East Europe and Central Asia Union of PLWH (ECUO)
Era of Mercy, Ukraine
E.V.A. Russian Network of Women Affected by HIV
Global Health Impact
The Global Network of People Living with HIV (GNP+)
Guli Surkh, Tajikistan
Health Action International (HAI)
Health GAP (Global Access Project)
Interagency Coalition on AIDS and Development (ICAD)
International Treatment Preparedness Coalition in Eastern Europe and Central Asia (ITPCru)
Kindernothilfe, Germany
Médecins Sans Frontières (MSF) Access Campaign
MEZIS
Pan African Positive Women's Coalition, Zimbabwe
Small Heart with Art, Ukraine
STOPAIDS
TB Proof
Thai Network of People Living with HIV/AIDS (TNP+)
The Sentinel Project on Pediatric Drug-Resistant Tuberculosis
The Republican Public Association “People Plus”
Treatment Action Campaign
Treatment Action Group (TAG)
Yolse, Santé Publique & Innovation
Youth Association for Development (YAD) Pakistan
Zimbabwe Women Living With HIV National Forum